Difference between revisions of "2017 Feb Taeyoung Lab note"
(→2017.2.22) |
(→2017.2.22) |
||
| Line 95: | Line 95: | ||
==2017.2.22== | ==2017.2.22== | ||
| + | ===LIPs PCR=== | ||
[[File:2017022201.jpg|frameless|2017.2.22 LIPs-13a Gradient using CS-14 and W82 Tm:50-65 130ml gel 1% Agar 100v 1h]] | [[File:2017022201.jpg|frameless|2017.2.22 LIPs-13a Gradient using CS-14 and W82 Tm:50-65 130ml gel 1% Agar 100v 1h]] | ||
| Line 104: | Line 105: | ||
1% Agar 100v 1h | 1% Agar 100v 1h | ||
| + | |||
| + | ===Muay GBS filtering and mapping=== | ||
| + | python run_trimming.py | ||
| + | python run_mapping.py | ||
| + | |||
| + | ===KaKs calculation using Jatropha species transcriptome=== | ||
| + | ====retrieve common gene between blastp and orthomcl in nocdhit result==== | ||
| + | /alima9002/63_backup/Jatropha/CDS/KaKs_nocdhit : python ret_common_gene_between_groups_and_blastp.py Jct.Jac.blastp.1e-10.na5.outfmt6 groups.txt Jct.Jac.blastp.1e-10.na5.outfmt6.grouped | ||
| + | ====KaKs calculation using genes from blastp only==== | ||
| + | /alima9002/63_backup/Jatropha/CDS/KaKs_only_blast : python run.kaks.py | ||
Latest revision as of 07:57, 22 February 2017
Contents |
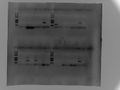
2017.2.2
PCR with LIP short primer
Ladder, 1-wild,1-cultivated,2-wild,2-cultivated order
LIP01-24
Last 4 bands are Tubulin control in CS-12,CS-14,KS-04,W82
1% agar, 100V, 1h, Tm:56,
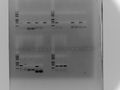
2017.2.3
PCR with LIP short primer
Ladder, 1-wild,1-cultivated,2-wild,2-cultivated order
SLIP01,02,03,06,07,09,13,14,15,16,20,24
Last 3 bands are Tubulin control in CS-12,CS-14,W82
1% agar, 100V, 1h, Tm:56,
2017.2.6
PCR with LIP short primer
Ladder, 1-wild,1-cultivated,2-wild,2-cultivated order
SLIP01,02,03,06,07,13,14,15,16,20,24
Last 3 bands are Tubulin control in CS-12,CS-14,W82
1% agar, 100V, 1h, Tm:56,
2017.2.7
PCR with LIP short primer
Ladder, 1-wild,1-cultivated,2-wild,2-cultivated order
2017.02.07 SLIP-08,12,13,20 with Wild and W82
Last 3 bands are Tubulin control in CS-12,CS-14,W82
1% agar, 100V, 1h, Tm:56,
Primer re-design
SLIP-13 and 16 is re-designed
python ~/py/LIP/6-1.manual_primer_design.py CS-14-contigs.fa.blastn.single_type.blastn.indel_candidates.read_evidence ../../CS-14/qvalue/CS-14-contigs.fa > CS-14.short.primer.fa.2candidates
2017.2.8~2.10
Field work
2017.2.13
LIPs-13 and 16 re-design
python ~/py/LIP/6-1.manual_primer_design.py CS-14-contia.blastn.single_type.blastn.indel_candidates.read_evidence ../../CS-14/qvalue/CS-14-contigs.fa > CS-14.short.primer.fa.2candidates
e-PCR
re-PCR -S ../../CS-14/qvalue/CS-14-contigs.hash -n1 -g1 CS-14.short.primer.fa.2candidates.sts -o CS-14.short.primer.fa.2candidates.sts.out
Primer order
OrthoMcl of Jatropha organ
all done
Draw venn diagram using Jatropha organ data
require(Vennerable)
Data <- read.table("group.txt.binary.parsed.rawinput.endosperm",col.names="")
Organ <- c("SG","LG","Y","B")
png(filename="Venn.endosperm.png",width=2000,height=2000)
plot(Venn(SetNames=Organ,Weight=as.vector(Data$X)),doWeights=F,type="ellipses")
dev.off()
OrthoMcl of Jatropha species
all done
Drawing venn diagram using Jatropha species data
done
BLASTP for Ks calculation among Jatropha species
193:/data2/alima90/Jatropha/KaKs_nocdhit$python blastp.py # blastp -db Jct.cds.fa.pep.complete.fa -evalue 1e-10 -outfmt 6 -num_threads 20 -num_alignments 5 -query Rco.cds.fa.pep.complete.fa -out Jct.Rco.blastp.1e-10.na5.outfmt6
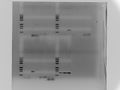
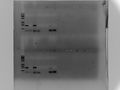
2017.2.20
LIPs-13a,16a with CS-14 and W82
Tm:56
1% agar 130ml gel 100v 1h
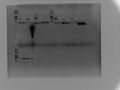
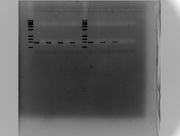
2017.2.22
LIPs PCR
LIPs-13a Gradient using CS-14 and W82
Tm:50-65
130ml gel
1% Agar 100v 1h
Muay GBS filtering and mapping
python run_trimming.py python run_mapping.py
KaKs calculation using Jatropha species transcriptome
retrieve common gene between blastp and orthomcl in nocdhit result
/alima9002/63_backup/Jatropha/CDS/KaKs_nocdhit : python ret_common_gene_between_groups_and_blastp.py Jct.Jac.blastp.1e-10.na5.outfmt6 groups.txt Jct.Jac.blastp.1e-10.na5.outfmt6.grouped
KaKs calculation using genes from blastp only
/alima9002/63_backup/Jatropha/CDS/KaKs_only_blast : python run.kaks.py